Stylized
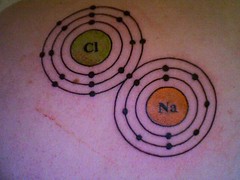
Good job on counting up the electrons and the ordering of the Bohr atomic shells in this tattoo, but to me the image suggests a valence bond relation, rather than an ionic bond appropriate for a compound of such widely varying electronegativities. That last electron does not want to have anything much to do with the sodium and is firmly in the camp of the chlorine ion.
If I were the wearer, which I'm not, it would bother me.
If I were the wearer, which I'm not, it would bother me.

2 comments:
OMG ouch!
Okay, that's definitely a covalent bond, as shown, not an ionic one.
Also the electron shell of the Cl- should be much larger than that of the Na+
So the tattoo artist isn't a chemist, nor the person wearing the tattoo... so presumably this will only bother a few geeks.
Anyone want to start a crowdsourcing site that lets experts in relevant fields 'fact-check' tattoos before people put them on their bodies?
Perhaps you'd like my other blog posting about a chemistry geek tattoo.
Well one has to allow a little bit of artistic license when using Bohr orbits like that instead of cloudlike space-filling atomic orbitals which I feel would make a poor artistic choice for this tattoo anyhow. So I'm willing to cut them some slack when it comes to scale.
Fact-checking would indeed help not only with technical lapses like this one but also the phenomenon of nonsense Chinese character tattoos. But I don't know whether it would stem the flood of non-anatomically-correct hearts inscribed on people's biceps.
Post a Comment